
Carbon dioxide acts as a greenhouse gas, trapping heat in Earth’s atmosphere. Carbon dioxide also is released when organic matter burns (including fossil fuels like oil or gas). The nucleus is orbited by a cloud of negatively charged electrons.Ĭarbon: The chemical element having the atomic number 6.Ĭarbon dioxide: (or CO 2) A colorless, odorless gas produced by all animals when the oxygen they inhale reacts with the carbon-rich foods that they’ve eaten.

Atoms are made up of a dense nucleus that contains positively charged protons and uncharged neutrons. Today, its roughly 1,400 scientists and engineers (and 1,000 students) conduct research across a broad range of fields, from biology and physics to materials science, energy development and climate studies.Ītmosphere: The envelope of gases surrounding Earth or another planet.Ītom: The basic unit of a chemical element.
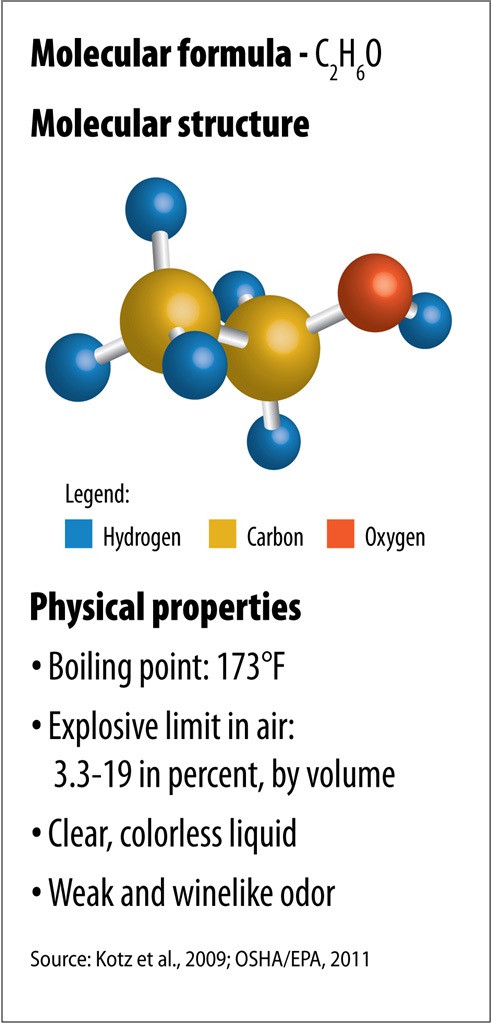
Department of Energy, outside of Chicago, Ill. “The truth is this is just the beginning of this long research journey.”Īrgonne National Laboratory: A federal laboratory owned by the U.S. That’s why the new study isn’t a final step, Liu says. They also hope to make other useful materials from carbon dioxide. The researchers are continuing to experiment with the new process. “It could be more complex than what is presented.” A closer look at the reaction is needed, she says. He says the new paper describes “a very interesting, innovative catalyst.”Īnna Klinkova also describes the new catalyst as “very cool.” Still, this chemist at the University of Waterloo in Canada remains a bit skeptical. Paul Kenis is a chemical engineer at the University of Illinois at Urbana-Champaign. All of these freed atoms now rearrange themselves to form ethanol (C 2H 6O). And the water - or H 2O - splits into hydrogen and oxygen atoms. The CO 2 now splits into carbon and oxygen atoms. Those clusters activate the catalyst, helping the CO 2 react with water. By firing powerful X-rays at them, Liu says his team could “monitor the chemical reaction as it happens.” Explainer: What is a catalyst? When electricity runs through the catalyst, groups of distant copper atoms join up, forming clusters of 3 or 4 atoms. Those copper atoms are spread out, as if social distancing. The new catalyst uses a bed of carbon to support copper atoms. Liu and his team described their work July 27 in Nature Energy. More than 90 percent of the final product is ethanol, he reports. And although chemical reactions often make many unwanted byproducts, this new one does not. With it, the voltage needed to start the new CO 2-to-ethanol reaction is far less than what’s needed to kickstart similar reactions, Liu says. A chemist at Argonne National Laboratory, in Lemont, Ill., he headed the team that developed this catalyst. Here, the group developed a brand-new catalyst, notes Di-Jia Liu. Such materials speed up a chemical reaction. In such circumstances, chemists often turn to a catalyst. To do that, the researchers first needed to break CO 2 into its building blocks. So making ethanol from CO 2 would be better for the environment But it offers a nice tradeoff: It helps that gasoline now burn more cleanly. Explainer: CO 2 and other greenhouse gasesĪdding ethanol reduces the energy in a gallon of gasoline. It’s the octane-boosting chemical added to most stocks of U.S. Wouldn’t it be great if that CO 2 pollution could instead by turned into a useful product? One team of researchers thinks it’s found a way to do just that. But too much of that gas has lately been driving an atmospheric fever. A new copper-based material might help chemists recycle an unwanted greenhouse gas into a desirable green fuel.Īs a greenhouse gas, carbon dioxide - or CO 2 - helps warm the atmosphere.
